The Glass method Prints can be taken on a variety of materials and should be stored in a cool dark place.
tin-foil, waxed paper, thin plastic disks that they use in jam making for covers are also a good medium. Paper can also be used but this involves soaking the paper in isopropanol before use and letting it dry.
The simplest way to take a print is to take a tall glass and some tin-foil that has been wiped with isopropanol to steralise it, place the foil on a sterile surface.
Using a sterile needle stick it the cap so that its not going to drop when you cut the stalk.
Cut the head from the stalk as close to the gills as you can but trying not to actually break the gills.
Either place the cap directly on the foil or I use a little chicken wire table to hold the print just off the surface which will help against contaminating the print.
Place the glass (I use a pint glass) over the cap and foil so that there isn't going to be a breeze blowing your spores all around the room. And leave for 24-36hrs.
The problem with using the glass method is contamination. The cap will use up the oxygen in the glass and begin to rot, also if you wish to take more than 1 print at a time it will soon take up your kitchen table ;)
Glovebox Method I have a simple glovebox (which I have described somewhere on these pages)that I use for taking prints and take them on transparent film used in jam making. These are ideal for examining your spores under your microscope and taking notes.
It is a lot easier taking multiple spore prints in a simple box and dramatically reduces the risk of the prints getting contaminated because of the larger volume of oxygen available.
I would stick to single strain multiple prints though as there is a danger of cross-spore contamination when taking prints of different strains in the same enclosed space.
i.e. p.mexicana and B+...the B+ will likely contaminate the p.mexicana print with any floating spores which will overpower the p.mexicana spores and you will end up with 2 B+ prints...could be annoying and expensive.
It is a good idea to thoroughly clean the glovebox when switching between cubes and other varieties of mushroom.
After the 24-36hr period the cap will have begun to shrink and you will be able (hopefully) to see the dropped spores on the material you have used. Print size and density will vary between caps and strains.
If using a glovebox make sure you put some zip-lock bags in there before you start your prints, it makes it easier to fold the print and pop it straight into the bag and not exposing it to the outside elements.
Lable and date each print so there is no confusion when adding it to your collection.
Your collection of mushroom prints will soon build up and you can swap prints with your friends which will build your collection even quicker. 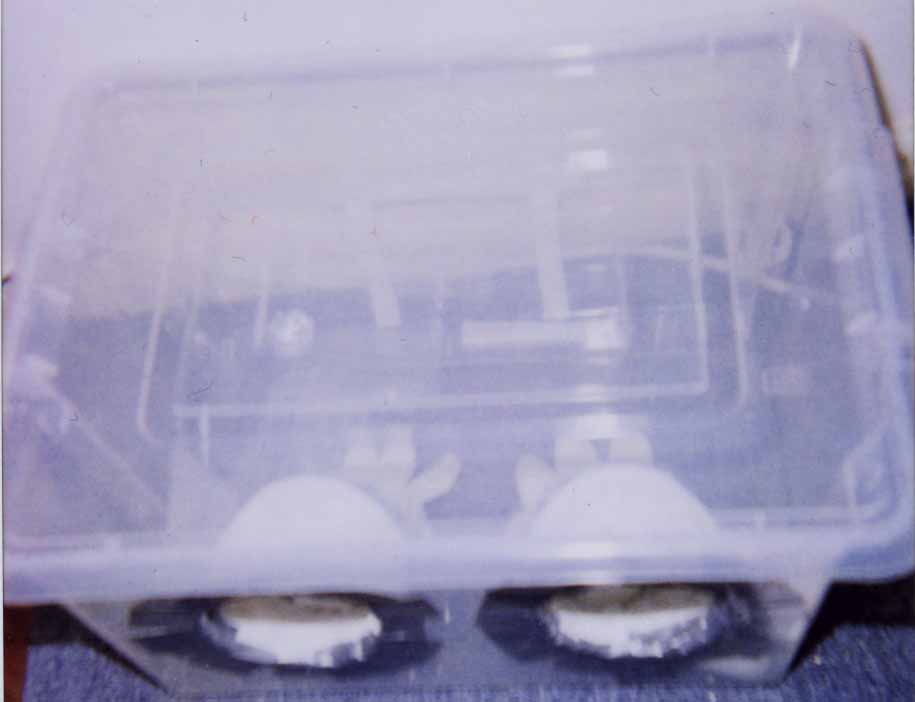
A very basic glovebox for taking prints/preparing syringes.

Viewed from a different angle with access via a resealable flap.

The jam jar covers are very cheap and I have found to be an excellent medium for spore prints. The spores come off very easily. These plastic covers do, like all mediums, need to be wiped with an isopropanol swab before use to ensure they are sterile.
|